Understanding composite endpoints: their possible strengths and weaknesses
Posted on 19th October 2023 by Sebastian Osorio Rico

During a cardiovascular consultation, you attend to a patient with obesity, hypertension, and dyslipidemia, but with no history of diabetes. The patient, concerned, raises the issue about the potentially fatal risk of diabetes in addition to his current conditions. Therefore, he inquires whether, apart from lifestyle modifications and usual care, there is any pharmacological treatment that can help prevent diabetes or reduce the risk of cardiovascular death.
Based on the DREAM study, you wonder if rosiglitazone might be effective in preventing type 2 diabetes in high-risk individuals. Although a significant 60% reduction in the primary outcome is observed, you realize that this includes both the incidence of diabetes and death. However, a more detailed analysis shows that the reduction is mainly attributed to a decrease in the incidence of diabetes and not in deaths. Given these findings, the question arises: Is incorporating these results into daily clinical practice prudent? Join me in exploring this type of outcome further.
What are composite endpoints?
Composite endpoints (also called composite outcomes) combine two or more simple outcomes, known as component outcomes, into a single variable. Patients who experience any of these components are considered to have reached the clinical endpoint. It is important not to confuse composite endpoints with primary and secondary outcomes. The primary outcome is the main objective of a study which the researcher wishes to assess, and this can be either simple or composite.
One of the most recognized composite endpoints in the medical literature is “Major Adverse Cardiac Events” (MACE), frequently assessed in cardiovascular clinical trials. This typically includes events such as non-fatal stroke, non-fatal myocardial infarction, and death due to cardiovascular causes, among others. In other fields, like oncology, the composite endpoint can sometimes be less direct, especially when the variable in question is continuous. For example, the term “progression-free survival” refers to the absence of two events: death and disease progression. This is another example of a composite endpoint.
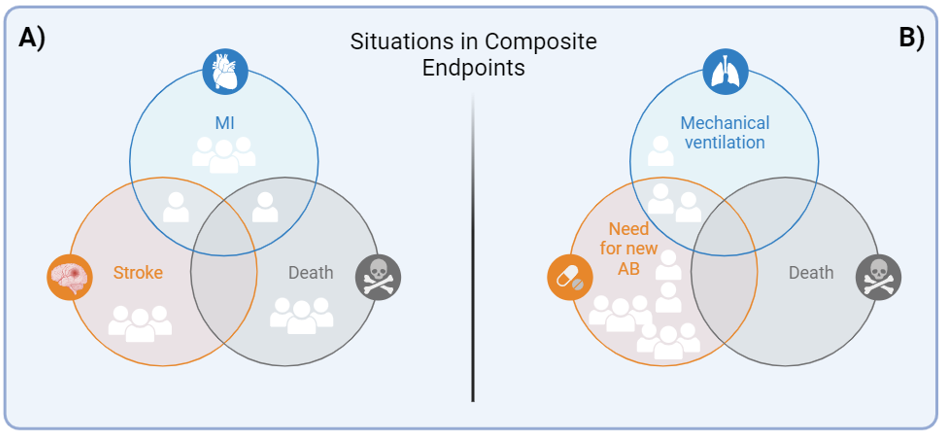
Let’s visualize two distinct clinical trials, both are placebo-controlled and with 100 patients in each study arm (Fig 1). In Clinical Trial A, the purpose is to determine if a new antiplatelet agent can reduce the incidence of MACE in patients with a history of MI. It is relevant to observe that, in total, 13 major cardiovascular events occurred, with certain patients experiencing more than one event. However, this figure is reflected in 11% of MACE, as it is recorded from the first presented episode. It is crucial to emphasize that, in general, all component outcomes show a similar distribution and clinical relevance. On the other hand, in Clinical Trial B, the placebo arm of a clinical trial is examined with the aim of assessing whether the use of antibiotics in patients with COPD exacerbations, but without sputum purulence, could influence clinical outcomes. A total of 13 events were recorded in the placebo arm, resulting in a composite endpoint of 11%. If the intervention group shows a lower incidence in this composite endpoint, it could lead to a misguided interpretation, suggesting that the intervention affects mortality. It’s vital to recognize that this difference might be mainly attributed to an elevated need to initiate new antibiotic treatment.
Benefits of using composite endpoints
It would be a challenge to statistically demonstrate a reduction in rare adverse events, such as bronchoaspiration when using second-generation supraglottic devices compared to first-generation ones. It is estimated that we would need a sample of tens of thousands of patients to achieve the necessary statistical power to evidence the superiority of the second-generation devices. When an outcome is rare, using composite endpoints reduces the required sample size. This often allows for a shorter follow-up period and lowers the study costs.
Treatments targeted at a particular disease do not always affect a single outcome; they can influence several simultaneously. Considering this multidimensional perspective, it’s useful to employ composite endpoints. For example, in a patient with ST-segment elevation myocardial infarction, when opting for an antiplatelet treatment with P2Y12 inhibitors and selecting the ideal antiplatelet agent, it’s not only relevant to avoid mortality but also essential to prevent new cardiovascular events, bleeding, heart failure, and the need for further revascularizations.
Disadvantages and criticisms
Despite the inherent advantages of using composite endpoints, it’s essential to recognize their challenges. These outcomes, by combining multiple components, must ensure that each of these has similar clinical relevance and magnitude to prevent misleading or erroneous interpretations. There’s a risk that a single component might disproportionately influence the overall result, making a meticulous and well-founded interpretation by the healthcare professional imperative.
Similarly, when the component outcomes have variations in their clinical importance, this can create complications in both the interpretation of results and effective communication with patients and other medical professionals. It also limits the possibility of systematically comparing different clinical trials unless this composite endpoint is reproducible and already defined and widely accepted based on an accepted expert consensus.
It is important to understand that if one of the component outcomes is manifested, it automatically means the composite endpoint has occurred. Additionally, some patients may experience more than one component outcome, but these patients are recorded only once in the category of the present composite endpoint. This rule applies to both outcomes that are dichotomous, meaning they are categorized as ‘Present’ or ‘Not Present’, and to continuous variables, such as ‘progression-free survival’ and others alike.
Interpretation of composite endpoints
While there are no internationally recognized or specifically adopted guidelines for the analysis of these types of outcomes, the following section will present some suggestions and recommendations, taking into account the criteria established in Guyatt’s User’s Guide to the Medical Literature published by JAMA.
Regardless of the external and internal validity analyses that characterize each study, it is essential to maintain a clear definition and meticulous selection of the components that make up the composite endpoint. It is imperative to consider the inherent clinical relevance of each component and ensure that they all share a coherent underlying biology.
When assessing fluctuations in the relative risk of the main outcome, it’s crucial to corroborate that variations in the estimates of said risk are coherent and consistent among the various components of the composite endpoint. That is, if a reduction in relative risk is identified in the composite endpoint, this should be similarly reflected in the decrease in risk for each of its individual components. If these criteria are not adhered to, it is recommended to examine and analyze each component outcome independently in detail.
Conclusions
Composite endpoints have gained prominence in research, primarily due to their methodological and economic benefits. However, when faced with the task of analyzing and interpreting such outcomes, it is imperative to approach them with meticulousness, as they have certain limitations that require thorough scrutiny of each of the components that constitute them.
References
- Guyatt G, Rennie D, Meade MO, Cook DJ. eds. Users’ Guides to the Medical Literature: A Manual for Evidence-Based Clinical Practice, 3rd ed. McGraw Hill; 2015.
- Baracaldo-Santamaría D, Feliciano-Alfonso JE, Ramirez-Grueso R, Rojas-Rodríguez LC, Dominguez-Dominguez CA, Calderon-Ospina CA. Making Sense of Composite Endpoints in Clinical Research. J Clin Med. 2023 Jun 29;12(13):4371.
- Armstrong PW, Westerhout CM. Composite End Points in Clinical Research: A Time for Reappraisal. Circulation. 2017 Jun 6;135(23):2299-2307.