Crossover trials: what are they and what are their advantages and limitations?
Posted on 7th September 2020 by Philip Heesen
Crossover trials are trials in which participants do not only receive one intervention, but multiple, and the effect of the interventions are measured on the same individuals. It is also described as participants receiving a sequence of interventions.
To make this concept less abstract, let us look at an example…
A researcher wants to compare the efficacy of the drug Valsartan with Iosartan, both given to treat high blood pressure. However, the researcher fears there might be a substantial risk of confounding (this beginner’s guide to confounding will help you understand this concept). In order to minimize this risk, she decides to make use of a crossover trial.
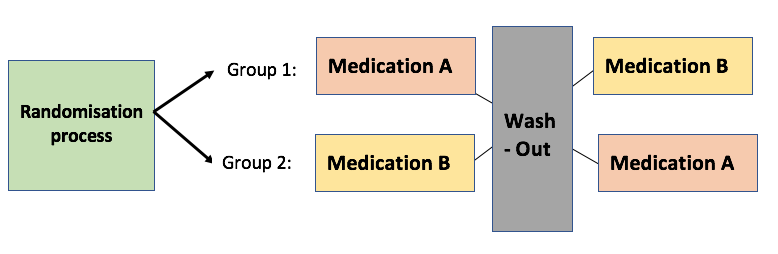
She recruits 120 participants into her study and randomly allocates them into 60 participants in group A and 60 participants in group B. At first, participants in group A will receive Valsartan for two weeks in order to treat their high blood pressure, and group B will receive Iosartan for a period of two weeks. After that, there will be a wash-out period of 4 weeks in which the study participants will not receive Iosartan or Valsartan. After that, group A will receive Iosartan for two weeks and group B will receive Valsartan for two weeks.
The researcher measures her primary outcome (reduction in mean blood pressure) twice: the first time after group A received Valsartan and a second time after group A received Iosartan.
This example easily explains the main steps that are followed during a crossover trial of the AB/BA type. The AB/BA model is the simplest type of crossover trial. At first, participants of one group will receive medication A and after a wash-out period, participants of the same group will receive medication B. The same applies to the second study group, but the other way around. Extensions to this form include the ABC/CBA/BCA regimens.
Advantages
By using a crossover trial in order to compare several interventions, a researcher can minimize the risk of confounding because all interventions are measured on the same participants. One can say that study participants serve as their own control. This leads to another advantage which is less study participants are required compared to a standard parallel randomized controlled trial (RCT). Reduction of sample size is consistent with the principle in medical research to use resources wisely. Furthermore, blinding of study participants can be maintained and statistical tests assuming randomization can be used.
Limitations
This design sounds very appealing, however there are various limitations that need to be considered:
- Crossover trials can only be conducted when the disease persists for a longer period of time, hence, crossover trials are mostly used in studying chronic diseases. There are some short-term illnesses or acute conditions that might be cured once they are treated and there are treatments that will have a permanent effect (i.e. surgery) on the patient. It is usually not possible to perform crossover trials in such cases.
- There is a great risk for aliasing. This term describes the risk that there might be a carry-over from the effect of the previous intervention on to the effect of the next intervention thereby altering results. Therefore, the treatment effect might not only be due to the treatment itself, but also due to interactions between treatment and study-period/sequence. Statistical tests have been suggested in order to test the carry-over risk, but a great chance of a type-II error (falsely accepting the null hypothesis, here: falsely accepting the hypothesis that there is no interaction between treatment and study-period or study-group) persists.
- It is difficult to estimate the time required in order for the intervention to be fully washed-out. While it might be relatively simple to estimate the wash-out period when the intervention is given as a drug looking at the half-life of the studied medications, things become a lot trickier when the interventions include psychological therapies, for example.
- In contrast to parallel designs, crossover trials consist of two study periods. This means that they usually take up more time, and statistical analysis can be more complicated if participants do not complete all stages of the trial.
Conclusions
In conclusion, crossover trials are a good study design that can be used to efficiently compare interventions on as few participants as possible when studying chronic diseases. However, many requirements (low risk of carry-over, wash-out period etc.) must be met and therefore it is not used as often as a parallel RCTs.




No Comments on Crossover trials: what are they and what are their advantages and limitations?
This is easy and best way to explain the concept
14th August 2023 at 8:56 amExcellent explaination !
9th October 2022 at 3:10 pmIn a rct cross over study ,two study group and one control group..what will be the design.and how the research question formed?
28th June 2021 at 6:25 am