An introduction to different types of study design
Posted on 6th April 2021 by Hadi Abbas

Study designs are the set of methods and procedures used to collect and analyze data in a study.
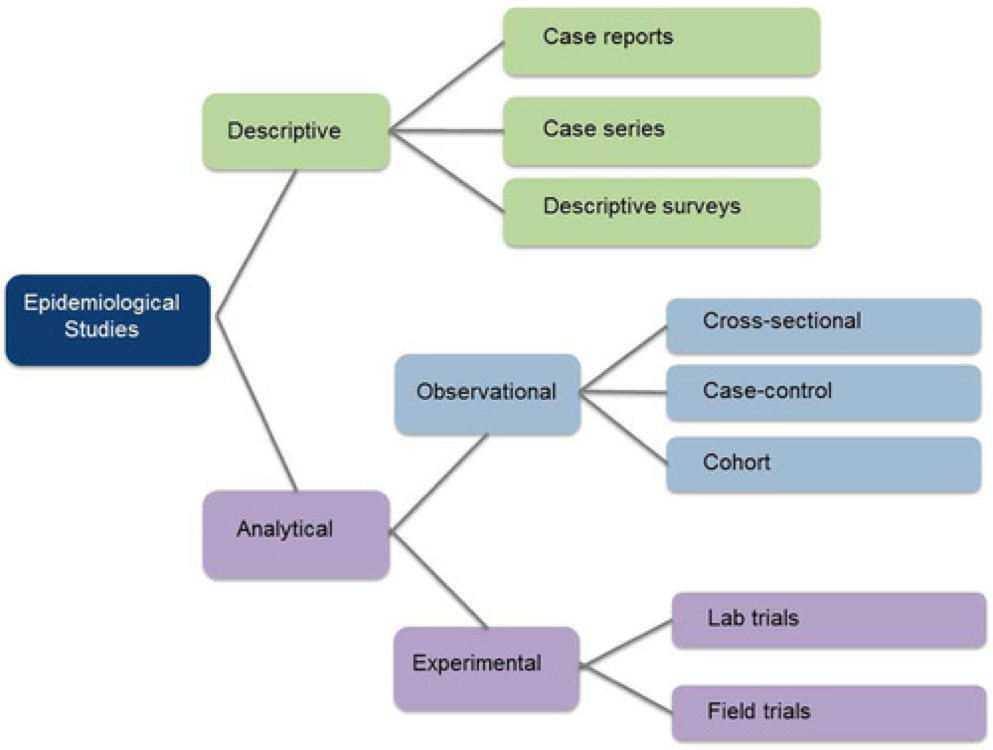
Broadly speaking, there are 2 types of study designs: descriptive studies and analytical studies.
Descriptive studies
- Describes specific characteristics in a population of interest
- The most common forms are case reports and case series
- In a case report, we discuss our experience with the patient’s symptoms, signs, diagnosis, and treatment
- In a case series, several patients with similar experiences are grouped.
Analytical Studies
Analytical studies are of 2 types: observational and experimental.
Observational studies are studies that we conduct without any intervention or experiment. In those studies, we purely observe the outcomes. On the other hand, in experimental studies, we conduct experiments and interventions.
Observational studies
Observational studies include many subtypes. Below, I will discuss the most common designs.
Cross-sectional study:
- This design is transverse where we take a specific sample at a specific time without any follow-up
- It allows us to calculate the frequency of disease (prevalence) or the frequency of a risk factor
- This design is easy to conduct
- For example – if we want to know the prevalence of migraine in a population, we can conduct a cross-sectional study whereby we take a sample from the population and calculate the number of patients with migraine headaches.
Cohort study:
- We conduct this study by comparing two samples from the population: one sample with a risk factor while the other lacks this risk factor
- It shows us the risk of developing the disease in individuals with the risk factor compared to those without the risk factor (RR = relative risk)
- We may approach this study by 2 longitudinal designs:
-
- Prospective: we follow the individuals in the future to know who will develop the disease
- Retrospective: we look to the past to know who developed the disease (e.g. using medical records)
-
- This design is the strongest among the observational studies
- For example – to find out the relative risk of developing chronic obstructive pulmonary disease (COPD) among smokers, we take a sample including smokers and non-smokers. Then, we calculate the number of individuals with COPD among both.
Case-Control Study:
- We conduct this study by comparing 2 groups: one group with the disease (cases) and another group without the disease (controls)
- This design is always retrospective
- We aim to find out the odds of having a risk factor or an exposure if an individual has a specific disease (Odds ratio)
- Relatively easy to conduct
- For example – we want to study the odds of being a smoker among hypertensive patients compared to normotensive ones. To do so, we choose a group of patients diagnosed with hypertension and another group that serves as the control (normal blood pressure). Then we study their smoking history to find out if there is a correlation.
Experimental Studies
- Also known as interventional studies
- Can involve animals and humans
- Pre-clinical trials involve animals
- Clinical trials are experimental studies involving humans
- In clinical trials, we study the effect of an intervention compared to another intervention or placebo. As an example, I have listed the four phases of a drug trial:
I: We aim to assess the safety of the drug (is it safe ?)
II: We aim to assess the efficacy of the drug (does it work ?)
III: We want to know if this drug is better than the old treatment (is it better ?)
IV: We follow-up to detect long-term side effects (can it stay in the market ?)
- In randomized controlled trials, one group of participants receives the control, while the other receives the tested drug/intervention. Those studies are the best way to evaluate the efficacy of a treatment.
Finally, the figure below will help you with your understanding of different types of study designs.
References (pdf)
You may also be interested in the following blogs for further reading:
An introduction to randomized controlled trials
Case-control and cohort studies: a brief overview
Cohort studies: prospective and retrospective designs
Prevalence vs Incidence: what is the difference?



No Comments on An introduction to different types of study design
you are amazing one!!
18th January 2024 at 1:10 amif I get you I’m working with you!
I’m student from Ethiopian higher education. health sciences student
Very informative and easy understandable
26th November 2023 at 5:07 pmYou are my kind of doctor. Do not lose sight of your objective.
3rd November 2023 at 6:05 pmWow very erll explained and easy to understand
3rd August 2023 at 9:34 amI’m Khamisu Habibu community health officer student from Abubakar Tafawa Balewa university teaching hospital Bauchi, Nigeria, I really appreciate your write up and you have make it clear for the learner. thank you
24th May 2023 at 12:31 pmwell understood,thank you so much
15th May 2023 at 11:31 pmWell understood…thanks
1st February 2023 at 5:21 pmSimply explained. Thank You.
20th October 2022 at 10:38 amThanks a lot for this nice informative article which help me to understand different study designs that I felt difficult before
8th August 2022 at 9:40 pmThat’s lovely to hear, Mona, thank you for letting the author know how useful this was. If there are any other particular topics you think would be useful to you, and are not already on the website, please do let us know.
21st September 2022 at 10:53 amDear sir
it is very informative and useful.
thank you
4th August 2022 at 10:33 amstatistician
Fabulous to hear, thank you John.
21st September 2022 at 10:53 amThanks for this information
27th July 2022 at 3:46 pmThanks so much for this information….I have clearly known the types of study design
27th July 2022 at 3:44 pmThanks
That’s so good to hear, Mirembe, thank you for letting the author know.
21st September 2022 at 10:54 amVery helpful article!!
14th July 2022 at 8:06 pmU have simplified everything for easy understanding
I’m a health science major currently taking statistics for health care workers…this is a challenging class…thanks for the simified feedback.
1st April 2022 at 7:57 amThat’s good to hear this has helped you. Hopefully you will find some of the other blogs useful too. If you see any topics that are missing from the website, please do let us know!
4th April 2022 at 9:47 amHello. I liked your presentation, the fact that you ranked them clearly is very helpful to understand for people like me who is a novelist researcher. However, I was expecting to read much more about the Experimental studies. So please direct me if you already have or will one day. Thank you
27th December 2021 at 3:29 amDear Ay. My sincere apologies for not responding to your comment sooner. You may find it useful to filter the blogs by the topic of ‘Study design and research methods’ – here is a link to that filter: https://s4be.cochrane.org/blog/topic/study-design/ This will cover more detail about experimental studies. Or have a look on our library page for further resources there – you’ll find that on the ‘Resources’ drop down from the home page.
However, if there are specific things you feel you would like to learn about experimental studies, that are missing from the website, it would be great if you could let me know too. Thank you, and best of luck. Emma
24th February 2022 at 2:50 pmGreat job Mr Hadi. I advise you to prepare and study for the Australian Medical Board Exams as soon as you finish your undergrad study in Lebanon. Good luck and hope we can meet sometime in the future. Regards ;)
9th December 2021 at 11:41 pmYou have give a good explaination of what am looking for. However, references am not sure of where to get them from.
8th December 2021 at 1:33 am